“Understanding the brain in all its complexity is impossible for any group to accomplish in isolation.”
-Arthur Toga, Director
We’ve built a diverse team of neurobiologists, mathematicians, and computer scientists, and a worldwide network of collaborators sharing data. Our goal is to increase the pace of discovery in neuroscience by better understanding how the brain works when it’s healthy and what goes wrong in disease.
About LONI
Our facility houses two advanced Magnetic Resonance Imaging scanners for data acquisition: a Magnetom Prisma 3T and a Magnetom Terra 7T.
Learn moreLONI’s onsite data center features state-of-the-art security technology and can store more than four petabytes of brain imaging data.
Learn moreOur Scientific Visualization Group creates elegant maps and animations to illustrate brain structure and function.
Learn moreLatest News
Latest News
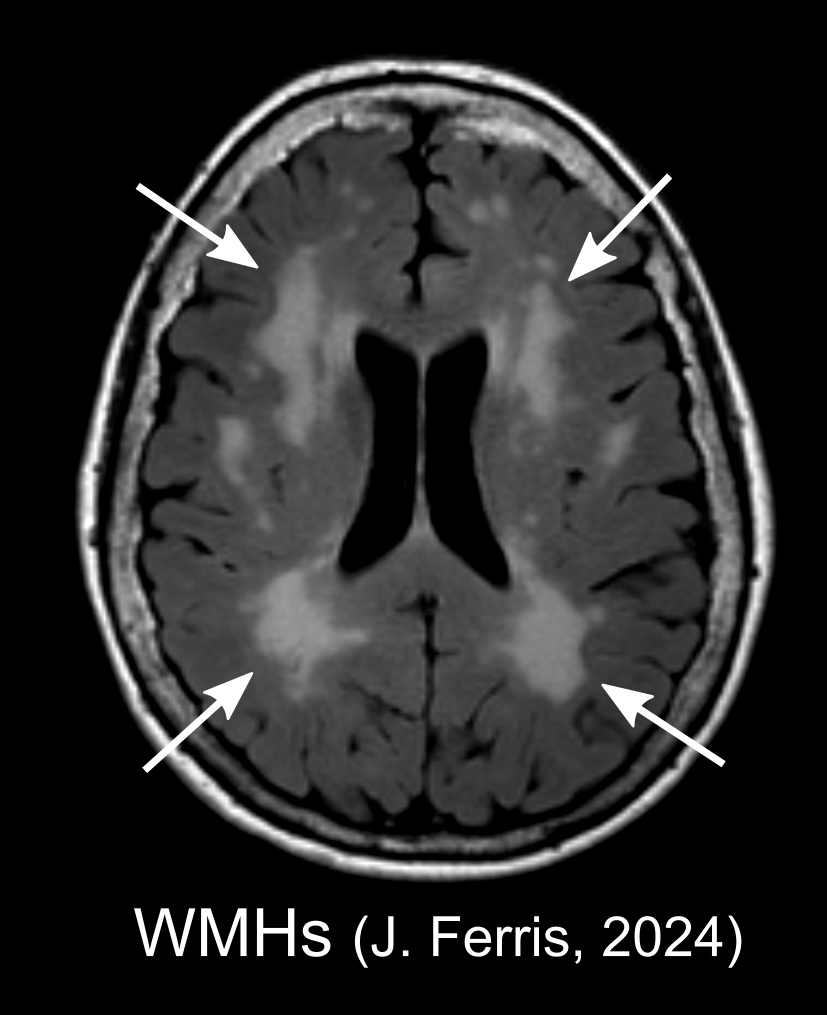
05/03/2024
A groundbreaking study published by a USC-led international consortium offers new insights into stroke recovery, revealing that age-related changes in brain white matter significantly affect how well individuals recover motor abilities following a stroke.
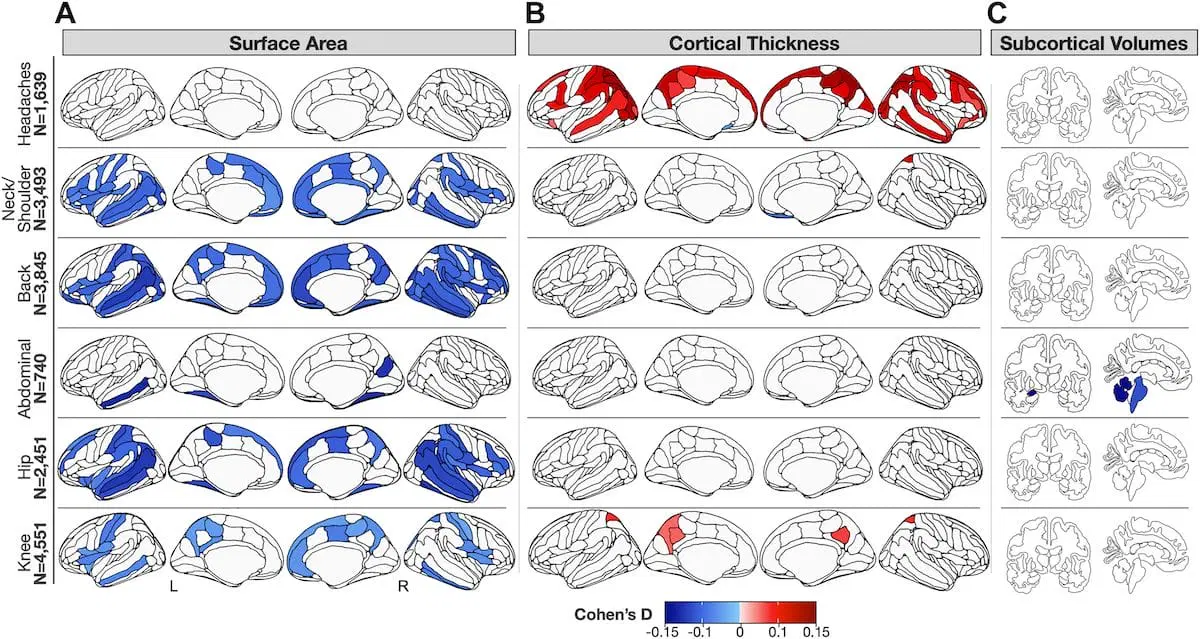
04/23/2024
A new neuroimaging study pinpoints differences in gray matter in those with chronic pain, especially multisite pain, compared to healthy individuals. Those who reported head pain showed unique differences compared to other chronic pain types.

04/16/2024
A new study led by Neda Jahanshad, PhD, is set to illuminate the underexplored domain of brain aging and risk for Alzheimer’s disease and related dementias (ADRD) among Middle Eastern and North African (MENA) adults in the US.
02/14/2024
Uniting experts in artificial intelligence, psychiatry, and neuroimaging, the ENIGMA Consortium will apply AI to brain scans of people with depression in the first global analysis for new ways to predict which patients will respond best to a promising new treatment.